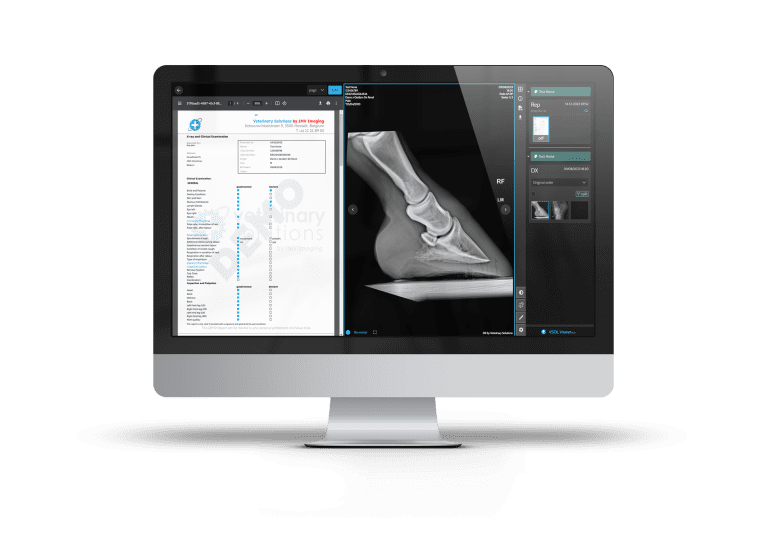
ConnectDR
The latest generation of DR X-Ray with slim and agile panel detectors, innovative acquisition software, and the capability to fully integrate your outstanding images into a truly connected imaging storage and distribution architecture. A variety of panel types to meet all the needs of equine, small animal or indeed exotic/wild-life vets.
Ultrasonography for Vet Nurses (Online Course + Practical Session with Jack Pye
Aimed at all skill levels, we will guide you through everything you need to know to become competent at ultrasonography. From patient preparation and using your own machine to how ultrasound is applied to our patients, we will get you confidently performing point-of-care ultrasound examinations of the thorax and abdomen.

The veterinary world is ever-changing and pet owners have never had higher expectations from their practices. We, at IMV imaging, can provide your team with the right tools to carry out more efficient imaging, through innovative equipment, expert resource and support, and education through our CPD courses.

All of our equipment is designed and manufactured in-house by our team of engineers or sourced through our relationships with the world’s leading veterinary healthcare and imaging providers.
Innovators in veterinary imaging
Through the years, we have chosen to be pioneers of industry. IMV imaging developed the first imaging product of its kind. Water resistant, wearable ultrasound scanners that allowed our customers to view their scanning images through a head mounted display. Since the product’s release we have continued to utilise our team of experts and global relationships to bring the best and most robust equipment to the veterinary industry.

Capturing and interpreting diagnostic images takes skill and confidence, which is why learning has always been at the heart of IMV.